
McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson
 |
Brought to you by: McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson |
|||
|
|
|
| Bioorganic
& Medicinal Chemistry Letters Volume 13, Issue 19, 16 October 2003, Pages 3297-3300 |
| ||||
|
|
| ||||
Synthesis of pyrimidinopyridine–triazene conjugates targeted to abl tyrosine kinase
Zakaria Rachid, Athanasia Katsoulas, Fouad Brahimi and Bertrand Jacques
Jean-Claude![]() ,
, ![]()
Abstract
The synthesis and abl tyrosine kinase inhibitory activities of alkyltriazenes
conjugated to phenylaminopyrimidines are described. Significant abl inhibitory
activities were observed only when a benzamido spacer was inserted between the
1,2,3-triazene chain and the 2-phenyaminopyridopyrimidine moiety.
Graphical Abstract
The synthesis and abl tyrosine kinase inhibitory activities of alkyltriazenes
conjugated to phenylaminopyrimidines are described. Significant abl inhibitory
activities were observed only when a benzamido spacer was inserted between the
1,2,3-triazene chain and the 2-phenyaminopyridopyrimidine moiety.
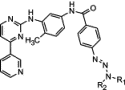 |
Display Full Size version of this image (1K) |
The alkyltriazenes represent one of the most potent classes of cytotoxic
drugs used in the clinic in the chemotherapy of many cancers including melanoma,
gliomas and leukemias.[1
and 2]
Dimethyltriazenes have superior stability when compared with monoalkyltriazenes.
[3]
They require metabolic activation to generate the cytotoxic monoalkyltriazene.
[4
and 5]
As outlined in Scheme
1, the clinical drug dacarbazine has been shown to be oxidized in vivo to
its corresponding hydroxymethyl metabolite which following loss of formaldehyde
gives the potent methyltriazenylimidazole-4-carboxamide (MTIC), a
monoalkyltriazene that is further hydrolyzed to a methyldiazonium species and
5-aminoimidazole-4-carboxamide.[6
and 7]
Recently, the cyclic triazene temozolomide (TEM), a stable prodrug of MTIC was
developed that does not require metabolic oxidation to generate the cytotoxic
species. [8]
TEM is now used in the clinic in the therapy of many malignancies including
melanoma and brain tumours. [2
and 9]
However, the toxicity of these compounds which is believed to be associated with
their lack of tumor selectivity, is a major deterrent for their use in cancer
therapy. Despite the significant potency of triazenes in the clinic, little
effort is directed at ameliorating their tumor selectivity.
.gif) |
Display Full Size version of this image (3K) |
Display Full Size version of this image (4K) Scheme 1. Metabolic activation of dacarbazine.
Recently, we developed a novel strategy termed ‘Combi-Targeting’ that seeks to synthesize chimeric molecules designed to simultaneously block tyrosine kinase-mediated growth signaling while inducing cytotoxic DNA damage.[10, 11, 12 and 13] More importantly, the resulting molecules also termed ‘combi-molecules’ were designed to degrade to another inhibitor of the same tyrosine kinase under physiological conditions. This was expected to culminate into selective and sustained antiproliferative activity in cells whose growth depend on the targeted tyrosine kinase. Here, we report the first attempt to apply these principles to the targeting of bcr-abl, a tyrosine kinase proven to be the cause of chronic myelogenous leukemia (CML). [14 and 15]
The bcr-abl oncoprotein is a 210 KDa fusion protein that is believed to play an anti-apoptotic role and blockade of its abl kinase activity has now been shown to translate into significant antitumor activity in both mouse models and in the clinic.[14, 15 and 16] STI571 or Gleevec™, [16] one of the most potent in vitro and in vivo inhibitors of abl kinase has now been approved by the Food and Drug Administration for the therapy of CML. Unfortunately, clinical studies with Gleevec™ showed rapid development of resistance in CML patients. [16] To circumvent this problem, the combination of this potent c-abl inhibitor with cytotoxic agents has become an actively explored strategy. [17 and 18] Here we report the first attempt to combine these two major mechanisms of antitumour actions into single molecules termed: ‘combi-molecules’.
The design of our combi-molecules was first based on appending the triazene tail to the 2-phenylaminopyrimidinopyridine moiety (see square on the structure of STI571) since derivatives of the latter group retained significant bcr-abl kinase inhibitory activities.[19] Thus, the synthesis of the phenylaminopyrimidines 1 and 2 (Scheme 2) proceeded according to strategy described by Zimmermann et al.[20] Triazenes directly branched onto the 2-phenylaminopyrimidopyridine moiety were obtained by diazotization of 1 or 2 with nitrosonium tetrafluoborate in acetonitrile followed by addition of the desired amine and neutralization with triethylamine to give 3 and 4. Compound 11 was synthesized by addition of a 1/10 mixture of aqueous methylamine (40%)/aqueous formaldehyde (37%).
Display Full Size version of this image (9K) Scheme 2. (i) CH3CN, NOBF4, −5 °C; (ii) Et2O, Et3N, alkylamine or 1/10 mixture of aqueous methylamine (40%)/aqueous formaldehyde (37%); (iii) p-nitrobenzoylchloride, pyridine, DMAP, rt overnight; (iv) Fe, EtOH, H2O, AcOH, refluxing 4 h.
The abl inhibitory activities of these compounds were tested in an enzyme
immunosorbent assay (ELISA) using poly(![]() -glutamic acid-
-glutamic acid-![]() -tyrosine, 4/1)(PGT) as a substrate for isolated v-abl kinase.[20]
In this assay, STI571 showed an IC50=0.04 μM. This assay permits a
short 10-min drug exposure, thereby avoiding substantial degradation of the
triazenes prior to binding to the ATP site of abl kinase. Compounds
3 and 4 with the triazenes directly attached
to the phenylaminopyrimidine ring showed IC50 values in range 8–10
μM. In view of the rather weak activities of these triazenes, we decided to
insert the benzamide spacer to our conjugates as in
9–12. This was achieved by treating amines
1 and 2 with p-nitrobenzoyl chloride to
give 5 and 6 which were reduced with Fe in
ethanol to provide 7 and 8. Diazotization of
these amines (7–8) with nitrosonium
tetrafluoborate in acetonitrile followed by addition of the desired amine and
neutralization with triethylamine gave triazene conjugates
9–12. While 10[27]
and 12 were stable molecules, compounds 9 and
11 had half-lives of 16 and approximately 30 min, respectively,
as determined by spectrophotometric monitoring of disappearing peaks at 315 and
365 nm. Interestingly, the bcr-abl kinase inhibitory activities of the latter
compounds increased by more than 10-fold when compared with those of
1, 3 and 4 (see Table
1).
-tyrosine, 4/1)(PGT) as a substrate for isolated v-abl kinase.[20]
In this assay, STI571 showed an IC50=0.04 μM. This assay permits a
short 10-min drug exposure, thereby avoiding substantial degradation of the
triazenes prior to binding to the ATP site of abl kinase. Compounds
3 and 4 with the triazenes directly attached
to the phenylaminopyrimidine ring showed IC50 values in range 8–10
μM. In view of the rather weak activities of these triazenes, we decided to
insert the benzamide spacer to our conjugates as in
9–12. This was achieved by treating amines
1 and 2 with p-nitrobenzoyl chloride to
give 5 and 6 which were reduced with Fe in
ethanol to provide 7 and 8. Diazotization of
these amines (7–8) with nitrosonium
tetrafluoborate in acetonitrile followed by addition of the desired amine and
neutralization with triethylamine gave triazene conjugates
9–12. While 10[27]
and 12 were stable molecules, compounds 9 and
11 had half-lives of 16 and approximately 30 min, respectively,
as determined by spectrophotometric monitoring of disappearing peaks at 315 and
365 nm. Interestingly, the bcr-abl kinase inhibitory activities of the latter
compounds increased by more than 10-fold when compared with those of
1, 3 and 4 (see Table
1).
Table 1. Cytotoxicity and v-abl tyrosine kinase inhibitory activities for compounds 1–8 and 9–12
.gif)
ND, not determined.
These results can easily be rationalized in light of previous structure–activity relationships and X-ray structures of STI571 or its variants[21 and 22] co-crystallized with c-abl that showed a hydrogen bonding role for the benzamido moiety. Indeed, recent studies showed that in addition to hydrogen bonds between Met 318 amide backbone and the pyridine nitrogen, between the pyrimidine NH and the Thr 315 OH group, the amide bond of the benzamido moiety was involved in a donor–acceptor interaction wherein the oxygen of Glu 286 formed a hydrogen bond with the benzamide NH and the oxygen of the latter accepted a hydrogen bond from the amide NH of Asp 381. The monoalkyltriazenes were designed with the hope that the benzamide hydrogen could be replaced by the hydrogen of the non-conjugated tautomeric form as depicted in Figure 1. The lack of abl kinase inhibitory activity may be due to the notorious instability of this tautomer, the structure of which resembles the transition state for protolysis of monoalkyltriazenes.[23]
Display Full Size version of this image (2K) Figure 1. Predicted interactions of the non-conjugated tautomers of the monoalkyltriazenes with the active site of c-abl (dotted lines indicate possible hydrogen bonds).
Furthermore, the contribution of the piperazine ring to the potency of STI571 is now known. It is located in a partially hydrophobic pocket forming van der Waals interactions with Val 289, Phe 359 and Asp 381.22 Since these interactions would be unavailable in our conjugates, we did not expect our benzamide-containing conjugates to exhibit bcr-abl inhibitory activities superior or in the same range as STI571. However, surprisingly the dimethyltriazene showed activity in the submicromolar range (Table 1). More importantly, the hydroxymethyl derivative that differs from the dimethyltriazenes by only a hydroxy group was 5-fold less active. This may be explained by the disfavored orientation of the polar hydroxy group towards the partially hydrophobic pocket of abl.
To rationalize these results, we overlaid the MM2 optimized structures with the X-ray determined conformation[23] of c-abl-bound STI571, using the Chem3D software package. ( Fig. 2) The model showed that of all the triazenes (3–4, 9–12), the dimethyltriazene 10 adopted the conformation found the closest to that of STI571. The dimethyl group appears in an orientation where it may form, like the piperazinyl ring STI571, van der Waals interactions with the partially hydrophobic pocket of c-abl. The presence of the polar hydroxy group of 11 may decrease such interactions leading, as observed, to a lesser affinity for the ATP site. It is noteworthy that while the activity of 11 was 5-fold less than that of 10, it was 5–10-fold more potent than derivatives 1–4 (devoid of the benzamido spacer) and amines 7–8.
Display Full Size version of this image (8K) Figure 2. Overlay of STI571 with dimethyltriazene conjugate 10.
As outlined in Scheme 1, dialkyltriazenes like dacarbazine are known to be metabolized by oxidation of their methyl group to generate a methylol intermediate that is rapidly converted to the cytotoxic monoalkyltriazene. The latter ultimately decomposes to an aromatic amine and an alkylating agent. [6 and 7] Similarly, our dimethyltriazene 10 will require metabolic oxidation to generate its putative metabolite 11, the precursor of an alkylating species. Indeed, alkylating test using the p-nitrobenzylpyridine (NBP) assay,[25] showed that in contrast to dimethyltriazene 10, that is devoid of alkylating activity, hydroxymethyltriazene 11 strongly alkylated NBP (Table 1). The translation of these properties into cytotoxic activity was studied in K562 promyelocytic leukemia cells. Triazenes 3 and 4 showed significant cytotoxic activities; however, this must not be rationalized on the basis of their bcr-abl inhibitory activities in these cells as their IC50 values for abl TK inhibition were extremely high (8.6–10 μM). In contrast, dimethyltriazene 10 that does not generate methyldiazonium owes its strong cytotoxic effect solely to its abl kinase inhibitory activity. Interestingly, the hydroxymethyltriazene analogue 11, despite being a 5-fold weaker inhibitor of abl kinase than 10 was only 2-fold less cytotoxic than the latter. This may well be due to the combination of effects associated with the abl inhibitory activity and its ability to alkylate DNA. Indeed, it was found that compound 11 damage DNA in K562 cells (data not shown). Detailed mechanism of the cytotoxic effects of 10 and 11 are reported elsewhere.
From the current studies, we have identified a potent abl inhibitor of the
dimethyltriazene class that does not generate alkylating species in vitro and
synthesized its putative metabolite 11 that showed significant
bcr-abl tyrosine kinase inhibitory activity and alkylating activity comparable
with that of temozolomide, a prodrug of the dacarbazine-derived
monoalkyltriazene.[7]
Further studies are required in vivo to demonstrate the metabolic conversion of
10 to 11 and to establish the pharmacological
advantages of these novel chimeric molecular approach.
References
1. S.M. O'Reilly, E.S. Newlands, M.G. Glaser, M. Brampton, J.M. Rice-Edwards, R.D. Illingworth, P.G. Richards, C. Kennard, I.R. Colquhoun, P. Lewis and M.F.G. Stevens. Eur. J. Cancer 29A (1993), p. 940. Abstract | PDF (1325 K) | View Record in Scopus | Cited By in Scopus (0)
2. N.M. Blehen, E.S. Newlands, S.M. Lee, N. Thatcher, P. Selby, A.H. Cavert, G.J.S. Rustin, M. Brampton and M.F.G. Stevens. J. Clin. Oncol. 13 (1995), p. 910.
3. W.L. Bullerwell, L.R. MacGillivray, M.J. Zaworotko, K. Vaughan and D.E.V. Wilman. Acta Cryst. Sect. C (1995), p. 2624. Full Text via CrossRef
4. H.W. Manning, L.M. Cameron, R.J. LaFrance, K. Vaughan and R. Rajaman. Anti-Cancer Drug Des. 1 (1985), p. 37. View Record in Scopus | Cited By in Scopus (11)
5. L.M. Cameron, R.J. LaFrance, C.M. Hemens, K. Vaughan, R. Rajaraman, D.C. Chubb and P.M. Goddard. Anti-Cancer Drug Des. 1 (1985), p. 27. View Record in Scopus | Cited By in Scopus (14)
6. M.F.G. Stevens, J.A. Hickman, S.P. Langdon, D. Chubb, L. Vickers, R. Stone, F. Gaig, C. Goddard, N.W. Gibson, J.A. Slack, C. Newton, E. Lunt, C. Fizames and F. Lavelle. Cancer Res. 47 (1987), p. 5846. View Record in Scopus | Cited By in Scopus (147)
7. J.M. Reid, M.J. Kuffel, J.K. Miller, R. Rios and M.M. Ames. Clin. Cancer Res. 5 (1999), p. 2192. View Record in Scopus | Cited By in Scopus (30)
8. S.M. O'Reilly, E.S. Newlands, M.G. Glaser, M. Brampton, J.M. Rice-Edwards, R.D. Illingworth, P.G. Richards, C. Kennard, I.R. Colquhoun, P. Lewis and M.F.G. Stevens. Eur. J. Cancer 29A (1993), p. 940. Abstract | PDF (1325 K) | View Record in Scopus | Cited By in Scopus (153)
9. E.S. Newlands, M.F.G. Stevens, S.R. Wedge, R.T. Wheelhouse and C. Brock. Cancer Treat. Rev. 23 (1997), p. 35. SummaryPlus | Full Text + Links | PDF (6185 K) | View Record in Scopus | Cited By in Scopus (201)
10. S. Matheson, J. McNamee and B.J. Jean-Claude. J. Pharm. Exp. Ther. 296 (2001), p. 832. View Record in Scopus | Cited By in Scopus (29)
11. F. Brahimi, S. Matheson, J. McNamee, A. Tari and B.J. Jean-Claude. J. Pharm. Exp. Ther. 303 (2002), p. 238. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (22)
12. S.L. Matheson, J. McNamee and B.J. Jean-Claude. Chemother. Pharmacol. 51 (2003), p. 11. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (19)
13. Q. Qiu, F. Dudouit, S.L. Matheson, F. Brahimi, R. Banerjee, J.P. McNamee and B.J. Jean-Claude. Cancer Chemother. Pharmacol. 51 (2003), p. 1. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (19)
14. M.W. Deininger, S.A.D. Vieira, Y. Parada, L. Banerji, E.W.F. Lam, G. Peters, F.X. Mahon, T. Kohler, J.M. Goldman and J.V. Melo. Cancer Res. 61 (2001), p. 8005. View Record in Scopus | Cited By in Scopus (27)
15. Y. Kano, M. Akutsu, S. Tsunoda, H. Mano, Y. Sato, Y. Honma and Y. Furukawa. Blood 97 (2001), p. 1999. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (165)
16. L.J. Campell, C. Patsouris and K.C Rayeroux. Cancer Genet. Cytogenet. 139 (2002), p. 30.
17. R. Nimmanapalli, E. O'Bryan, M. Huang, P. Bali, P.K. Burnette, T. Loughram, J. Tepperberg, R. Jove and K. Bhalla. Cancer Res. 62 (2002), p. 5761. View Record in Scopus | Cited By in Scopus (61)
18. J. Topaly, W.J. Zeller and S. Fruehauf. Leukemia 15 (2001), p. 342. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (113)
19. J.T. Thiesing, S. Ohno-Jones, K.S. Kolibaba and B.J. Druker. Blood 96 (2000), p. 3195. View Record in Scopus | Cited By in Scopus (252)
20. J. Zimmermann, E. Buchdunger, H. Mett, T. Meyer, N. Lydon and B. Nicholas. Bioorg. Med. Chem. Lett. 7 (1997), p. 187. Abstract | Abstract + References | PDF (279 K) | View Record in Scopus | Cited By in Scopus (87)
21. J.D. Moyer, E.G. Barbacci, K. Iwata, L. Arnold, B. Boman, A. Cunningham, C. DiOrio, J. Doty, M.J. Morin, M.J. Moyer, M. Neveu, V.A. Pollak, L.R. Pustilnik, M.M. Reynolds, D. Sloan, A. Teleman and P. Miller. Cancer Res. 57 (1997), p. 4838. View Record in Scopus | Cited By in Scopus (324)
22. T. Schindler, W. Bornmann, P. Pellicena, W.T. Miller, B. Clarkson and J. Juriyan. Science 289 (2000), p. 1938. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (622)
23. B. Nagar, W.G. Bornmann, P. Pellicena, T. Schindler, D.R. Veach, T.D. Miller, B. Clarkson and J. Kuriyan. Cancer Res. 62 (2002), p. 4236. View Record in Scopus | Cited By in Scopus (272)
24. C.M. Hemens, H.W. Manning, K. Vaughan, R.J. LaFrance and Y. Tang. Can. J. Chem. 62 (1984), p. 741.
25. D. Vistica, P. Skehan, D. Scudiero, A. Monks, J.D. Pittam and M.R. Boyd. Cancer Res. 15 (1991), p. 2515. View Record in Scopus | Cited By in Scopus (193)
26. H. Nells, S.C. Alry and J.E. Sinshelmer. Anal. Chem. 54 (1982), p. 213. View Record in Scopus | Cited By in Scopus (5)
Experimental data for combi-molecule 10. Amine
8 (100 mg, 0.25 mmol) was dissolved in dry acetonitrile (10 mL)
and after cooling to −5 °C, a solution of nitrosonium tetrafluoroborate (60
mg, 0.5 mmol) suspended in acetonitrile was added. The clear solution was
stirred for 1 h at −5 °C and a mixture of ether (10 mL), water (2 mL),
triethylamine (0.5 mL) and dimethylamine (40%) (0.3 mL) was added dropwise. The
mixture was subsequently stirred at 0 °C for 2 h and the precipitate that
formed filtered and washed with ether/ethyl acetate to give 10
(75 mg, 66%) as a pure solid: mp 165 °C; APCI m/z 452.9
(MH+), 380 (M-dimethyltriazene, 76.7); 1H NMR
(400 MHz, DMSO-d6) δ 10.10 (s, 1H, CONH), 9.25 (s, 1H,
NH), 8.97 (s, 1H, ArH), 8.66 (d, 1H, J=4.8 Hz, ArH), 8.49–8.44 (m, 2H,
ArH), 8.05 (d, 1H, J=2.0 Hz, ArH), 7.93 (d, 2H, J=8.8 Hz, ArH),
7.52–7.46 (m, 2H, ArH), 7.4 (d, 2H, J=8.8 Hz, ArH), 7.18 (d, 2H,
J=8.4 Hz, ArH), 3.52 (br s, 3H, N(Me)2), 3.18 (br s, 3H,
N(Me)2), 2.21 (s, 3H, CH3); 13C NMR (100 MHz,
DMSO-d6) δ 165.5, 162.2, 160.1, 153.5, 152.0, 148.8, 138.4,
137.9, 135.1, 132.9, 131.7, 130.7, 129.4 (2C), 128.2, 124.5, 120.3 (2C), 117.9,
117.4, 108.2, 43.8, 36.8, 18.5
| Bioorganic
& Medicinal Chemistry Letters Volume 13, Issue 19, 16 October 2003, Pages 3297-3300 |
|
|
 |